Antibody data
- Antibody Data
- Antigen structure
- References [3]
- Comments [0]
- Validations
- Western blot [3]
- Immunocytochemistry [4]
- Other assay [4]
Submit
Validation data
Reference
Comment
Report error
- Product number
- PA5-28869 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- ZO-1 Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Recombinant protein fragment
- Description
- Recommended positive controls: HeLa, HCT116, HaCaT. Predicted reactivity: Mouse (99%), Rat (99%), Dog (100%).
- Reactivity
- Human
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 100 µL
- Concentration
- 0.98 mg/mL
- Storage
- Store at 4°C short term. For long term storage, store at -20°C, avoiding freeze/thaw cycles.
Submitted references Vardenafil increases intracellular accumulation of the most prevalent mutant cystic fibrosis transmembrane conductance regulator (CTFR) in human bronchial epithelial cells.
Inhibition of MicroRNA-155 Supports Endothelial Tight Junction Integrity Following Oxygen-Glucose Deprivation.
Resveratrol increases F508del-CFTR dependent salivary secretion in cystic fibrosis mice.
Dhooghe B, Bouzin C, Mottais A, Hermans E, Delion M, Panin N, Noel S, Leal T
Biology open 2020 Aug 25;9(8)
Biology open 2020 Aug 25;9(8)
Inhibition of MicroRNA-155 Supports Endothelial Tight Junction Integrity Following Oxygen-Glucose Deprivation.
Pena-Philippides JC, Gardiner AS, Caballero-Garrido E, Pan R, Zhu Y, Roitbak T
Journal of the American Heart Association 2018 Jun 26;7(13)
Journal of the American Heart Association 2018 Jun 26;7(13)
Resveratrol increases F508del-CFTR dependent salivary secretion in cystic fibrosis mice.
Dhooghe B, Bouckaert C, Capron A, Wallemacq P, Leal T, Noel S
Biology open 2015 Jun 19;4(7):929-36
Biology open 2015 Jun 19;4(7):929-36
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western blot analysis of ZO-1 using 30 µg of HCT116 lysate. Samples were loaded onto a 5% SDS-PAGE gel and probed with a ZO-1 polyclonal antibody (Product # PA5-28869) at a dilution of 1:5000.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
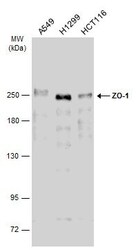
- Experimental details
- Western Blot analysis of ZO-1 was performed by separating 30 µg of various whole cell extracts by 5% SDS-PAGE. Proteins were transferred to a membrane and probed with a ZO-1 Polyclonal Antibody (Product # PA5-28869) at a dilution of 1:2000 and a HRP-conjugated anti-rabbit IgG secondary antibody.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Western Blot analysis of ZO-1 was performed by separating 30 µg of various whole cell extracts by 5% SDS-PAGE. Proteins were transferred to a membrane and probed with a ZO-1 Polyclonal Antibody (Product # PA5-28869) at a dilution of 1:2000 and a HRP-conjugated anti-rabbit IgG secondary antibody.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
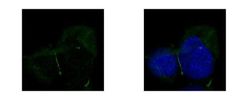
- Experimental details
- Immunofluorescent analysis of ZO-1 showing staining in the junction of A431 cells. A431 cells were fixed in ice-cold MeOH for 5 min and stained using a ZO-1 polyclonal antibody (Product # PA5-28869) diluted at 1:500. Blue: Hoechst 33342 staining.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
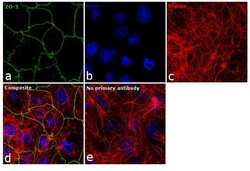
- Experimental details
- Immunofluorescence analysis of ZO-1 was performed using 90% confluent log phase Caco-2 cells. The cells were fixed with 4% paraformaldehyde for 10 minutes, permeabilized with 0.1% Triton™ X-100 for 15 minutes, and blocked with 1% BSA for 1 hour at room temperature. The cells were labeled with ZO-1 Rabbit Polyclonal Antibody (Product # PA5-28869) at 5 µg/mL in 0.1% BSA, incubated at 4 degree Celsius overnight and then labeled with Goat anti-Rabbit IgG (H+L) Superclonal™ Secondary Antibody, Alexa Fluor® 488 conjugate (Product # A27034) at a dilution of 1:2000 for 45 minutes at room temperature (Panel a: green). Nuclei (Panel b: blue) were stained with SlowFade® Gold Antifade Mountant with DAPI (Product # S36938). F-actin (Panel c: red) was stained with Rhodamine Phalloidin (Product # R415, 1:300). Panel d represents the merged image showing junctional localization. Panel e represents control cells with no primary antibody to assess background. The images were captured at 60X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
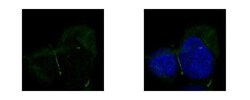
- Experimental details
- ZO-1 Polyclonal Antibody detects TJP1 protein at junction by confocal immunofluorescent analysis. Sample: A431 cells were fixed in ice-cold MeOH for 5 min. Green: TJP1 protein stained by ZO-1 Polyclonal Antibody (Product # PA5-28869) diluted at 1:500. Blue: Hoechst 33342 staining. [Images captured by Olympus FV10i Confocal Laser Scanning Microscope].
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
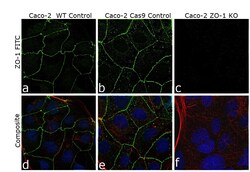
- Experimental details
- Knockout of ZO-1 was achieved by CRISPR-Cas9 genome editing using LentiArray™ Lentiviral sgRNA (Product # A32042, AssayID CRISPR533979_LV) and LentiArray Cas9 Lentivirus (Product # A32064). Immunofluorescence analysis was performed on wild type Caco-2 cells (panel a,d), Caco-2 Cas9 control cells (panel b,e) and Caco-2 ZO-1KO cells (panel c, f). Cells were fixed, permeabilized, and labeled with ZO-1 Polyclonal Antibody (Product # PA5-288698203, 3 µg/mL dilution), followed by Donkey anti-Rabbit IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor™ Plus 488 (Product # A32790, 1:2000). Nuclei (blue) were stained using ProLong™ Diamond Antifade Mountant with DAPI (Product # P36962), and Rhodamine Phalloidin (Product # R415, 1:300) was used for cytoskeletal F-actin (red) staining. Loss of signal (panel c,f) upon CRISPR mediated knockout (KO) confirms that antibody is specific to ZO-1 (green). The images were captured at 60X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 4. Quantification of CFTR protein by immunofluorescence in human bronchial epithelial cells. (A) Representative immunohistochemical labeling of CFTR (green) and zonula occludens (ZO-1, red) 24 h after incubation with DMSO (left) or 100 uM resveratrol (right) in wild-type 16HBE14o- (WT, top) or in CFBE41o- (CF, bottom) cultures. Nuclei (blue) stained by DAPI. Bar: 20 um. (B) Relative position corresponding to intensity of the CFTR signal quantified by morphometric analysis of cross-sectional profiles in individual ( n =8 cells per condition, randomly selected). (C) Average area under the fluorescence curves (+-s.e.m.) obtained in B normalized to that measured in DMSO-treated wild-type cells and expressed as a measure of total CFTR protein expression (* P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 3. Representative immunohistochemical images of CFTR in 16HBE14o - and in CFBE41o - cells. (A) Labelling of CFTR (green) and zonula occludens (Z0-1, red) 1 h after incubation with DMSO (A1 and A6), 10 uM 8-Br-cGMP (A3 and A8), 10 uM vardenafil alone (A2 and A7) or in combination with 1 uM PKA inhibitor (A5 and A10) or 1 uM PKG inhibitor (A4 and A9) in wild-type 16HBE14o - (left panel; 1-5) and in CFBE41o - cells (right panel; 6-10). In each picture, a small rectangle containing a region extending from the plasma membrane to the nucleus is enlarged (x4.5) in insert. Nuclei (blue) stained by DAPI. (B) Average area under the curves (AUC) obtained from the intensity of the CFTR signals quantified by morphometric analysis of cross-sectional profiles of individual cells ( n =9) and expressed as a measure of total CFTR protein expression (nuclei excluded). Each condition has been normalised to that measured in the control group (DMSO-treated 16HBE14o - cells). Data are expressed as mean+-s.e.m. Asterisks indicate levels of significance of between-group comparisons performed by ANOVA with post-hoc analysis made by using Student's t -test or Tukey-Kramer HSD test, as appropriate (* P
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4 Effect of microRNA-155 (miR-155) inhibition and overexpression on the endothelial cell junction proteins. Immunofluorescence microscopy (A) and Western blot analysis (B) were performed to detect the cellular distribution and expression levels of cell junction proteins in oxygen-glucose deprivation (OGD)/control inhibitor (OGD/IC), specific miR-155 inhibitor (OGD/I), and mimic (OGD/M) cells. A, Immunofluorescence staining for occludin, claudin-5, and zonula occludens protein-1 (ZO-1; red). Confocal microscopy images were acquired using a Zeiss LSM 800 confocal microscope. Bar: 10 mum. B, Representative immunoblots demonstrate the levels of occludin, claudin-5, ZO -1, and vascular endothelial (VE)-cadherin. In addition to cell junction proteins, Western blots were performed to assess expression of miR-155 direct target Rheb and RhoA bands. C, Graph demonstrates quantification of Western blot data obtained in OGD /I (black squares) and OGD /M (grey triangles) samples. Optical density of the protein bands was measured using ImageJ software, normalized to GAPDH density in every sample, and expressed as a percentage of the optical density calculated in the appropriate control OGD / IC and mimic control ( OGD / MC ) samples, respectively. n=4 (for OGD / IC and OGD / MC groups) and 6 (for OGD /I and OGD /M groups) independent experiments. Mann-Whitney (Wilcoxon) test was used to compare the relative protein levels between OGD /I and OGD / IC groups ( P =0.029). Error bars:
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6 Claudin-1 (CLDN1) and zonula occludens protein-1 (ZO-1) colocalize and interact in oxygen-glucose deprivation (OGD)/specific miR-155 inhibitor (OGD/I) cells. Double immunofluorescence staining of OGD /I cells with anti-CLDN1 (A) and anti- ZO -1 (B) antibodies. C and D, Colocalization maps automatically generated using Fiji software demonstrate colocalization (yellow) and no colocalization (red pixels) of 2 channels in the cells (C); enlarged details in (D) depict the degree of colocalization on the cell borders. Bar: 10 mum. E, Orthogonal projection of human primary brain microvascular endothelial cells from the OGD /I group, coimmunostained with anti-CLDN1 (green) and anti- ZO -1 (red) antibodies. 4',6-Diamidino-2-phenylindole staining was used to visualize nuclei (blue). Imaging was performed with a Zeiss LSM 800 confocal microscope using tile scan and Z stack image acquisitions. F, Cell lysates from the OGD /I and OGD /control inhibitor (OGD/IC) groups (L- OGD /I and L- OGD / IC ) were immunoprecipitated with anti- ZO -1 antibody. Immunoprecipitates ( IP - OGD /I and IP - OGD / IC ) were probed with antibodies against ZO -1, CLDN1, actin, phosphotyrosine ( pT yr), and phosphoserine ( pS er). G through I, OGD -subjected cells were transfected with CLDN 1 cDNA -containing vector ( OGD + CLDN 1) or empty vector ( OGD + EV ). G, expression of ZO -1 and CLDN1 was detected in OGD + CLDN 1 (lane 1), normoxic control ( NC ; lane 2), and OGD + EV (lane 3) samples. Actin
 Explore
Explore Validate
Validate Learn
Learn Western blot
Western blot