PA1-16601
antibody from Invitrogen Antibodies
Targeting: HIF1A
bHLHe78, HIF-1alpha, HIF1, MOP1, PASD8
 Western blot
Western blot Immunocytochemistry
Immunocytochemistry Immunoprecipitation
Immunoprecipitation Immunohistochemistry
Immunohistochemistry Chromatin Immunoprecipitation
Chromatin Immunoprecipitation Other assay
Other assayAntibody data
- Antibody Data
- Antigen structure
- References [31]
- Comments [0]
- Validations
- Immunocytochemistry [4]
- Immunohistochemistry [2]
- Other assay [21]
Submit
Validation data
Reference
Comment
Report error
- Product number
- PA1-16601 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- HIF1A Polyclonal Antibody
- Antibody type
- Polyclonal
- Antigen
- Other
- Description
- This antibody detects upregulation of HIF-1 alpha in hypoxic samples. Recommended positive controls: Human pancreas whole tissue lysate, COS-7 nuclear hypoxic induced cell lysate, human bone marrow whole tissue lysate (adult whole diabetes), HeLa hypoxic (CoCl2) cell lysate, HepG2 hypoxic (CoCl2) cell lysate, HeLa hypoxic cell Lysate, HepG2 hypoxic cell lysate, HIF-1 alpha knockout HeLa cell lysate
- Reactivity
- Human, Mouse, Rat, Canine, Goat, Hamster, Porcine, Rabbit, Zebrafish
- Host
- Rabbit
- Isotype
- IgG
- Vial size
- 100 μL
- Concentration
- 1 mg/mL
- Storage
- -20°C
Submitted references Oxygen microcapsules improve immune checkpoint blockade by ameliorating hypoxia condition in pancreatic ductal adenocarcinoma.
Modulatory Effects of Estradiol and Its Mixtures with Ligands of GPER and PPAR on MAPK and PI3K/Akt Signaling Pathways and Tumorigenic Factors in Mouse Testis Explants and Mouse Tumor Leydig Cells.
Immunorthodontics: PD-L1, a Novel Immunomodulator in Cementoblasts, Is Regulated by HIF-1α under Hypoxia.
Mitochondrial ROS drive resistance to chemotherapy and immune-killing in hypoxic non-small cell lung cancer.
LW1497, an Inhibitor of Malate Dehydrogenase, Suppresses TGF-β1-Induced Epithelial-Mesenchymal Transition in Lung Cancer Cells by Downregulating Slug.
Oxidation of Hemoglobin Drives a Proatherogenic Polarization of Macrophages in Human Atherosclerosis.
Obesity Impairs Embryonic Myogenesis by Enhancing BMP Signaling within the Dermomyotome.
Human Pluripotent Stem Cell-Derived Neural Progenitor Cells Promote Retinal Ganglion Cell Survival and Axon Recovery in an Optic Nerve Compression Animal Model.
Silicon Oxynitrophosphide Nanoscale Coating Enhances Antioxidant Marker-Induced Angiogenesis During in vivo Cranial Bone-Defect Healing.
An Important Role for DNMT3A-Mediated DNA Methylation in Cardiomyocyte Metabolism and Contractility.
Maternal erythrocyte ENT1-mediated AMPK activation counteracts placental hypoxia and supports fetal growth.
Oxygen Tension and the VHL-Hif1α Pathway Determine Onset of Neuronal Polarization and Cerebellar Germinal Zone Exit.
YAP1 and TAZ negatively control bone angiogenesis by limiting hypoxia-inducible factor signaling in endothelial cells.
Clinicopathological Analysis of HIF-1alpha and TERT on Survival Outcome in Glioblastoma Patients: A Prospective, Single Institution Study.
Enantioselective Synthesis of 8-Hydroxyquinoline Derivative, Q134 as a Hypoxic Adaptation Inducing Agent.
Chloramphenicol Induces Autophagy and Inhibits the Hypoxia Inducible Factor-1 Alpha Pathway in Non-Small Cell Lung Cancer Cells.
Macrophage-mediated delivery of light activated nitric oxide prodrugs with spatial, temporal and concentration control.
Ginsenoside Rg5 Inhibits Succinate-Associated Lipolysis in Adipose Tissue and Prevents Muscle Insulin Resistance.
Hypoxia-Inducible Factor-Dependent Expression of Angiopoietin-Like 4 by Conjunctival Epithelial Cells Promotes the Angiogenic Phenotype of Pterygia.
Hypoxia promotes primitive glycosaminoglycan-rich extracellular matrix composition in developing heart valves.
Metformin and resveratrol ameliorate muscle insulin resistance through preventing lipolysis and inflammation in hypoxic adipose tissue.
Inhibition of HIF Reduces Bladder Hypertrophy and Improves Bladder Function in Murine Model of Partial Bladder Outlet Obstruction.
The Neuroprotective Effect of Coumaric Acid on Spinal Cord Ischemia/Reperfusion Injury in Rats.
Effect of CoCl₂ on fracture repair in a rat model of bone fracture.
K63 linked ubiquitin chain formation is a signal for HIF1A degradation by Chaperone-Mediated Autophagy.
Snail Is a Direct Target of Hypoxia-inducible Factor 1α (HIF1α) in Hypoxia-induced Endothelial to Mesenchymal Transition of Human Coronary Endothelial Cells.
Wnt signaling directs a metabolic program of glycolysis and angiogenesis in colon cancer.
HIF-1α-induced HSP70 regulates anabolic responses in articular chondrocytes under hypoxic conditions.
Inhibition of mitochondrial complex III blocks neuronal differentiation and maintains embryonic stem cell pluripotency.
Response of small heat shock proteins in diabetic rat retina.
Superoxide induced by a high-glucose concentration attenuates production of angiogenic growth factors in hypoxic mouse mesenchymal stem cells.
Wu J, Wang X, Chen L, Wang J, Zhang J, Tang J, Ji Y, Song J, Wang L, Zhao Y, Zhang H, Li T, Sheng J, Chen D, Zhang Q, Liang T
Bioactive materials 2023 Feb;20:259-270
Bioactive materials 2023 Feb;20:259-270
Modulatory Effects of Estradiol and Its Mixtures with Ligands of GPER and PPAR on MAPK and PI3K/Akt Signaling Pathways and Tumorigenic Factors in Mouse Testis Explants and Mouse Tumor Leydig Cells.
Gorowska-Wojtowicz E, Duliban M, Kotula-Balak M, Bilinska B
Biomedicines 2022 Jun 12;10(6)
Biomedicines 2022 Jun 12;10(6)
Immunorthodontics: PD-L1, a Novel Immunomodulator in Cementoblasts, Is Regulated by HIF-1α under Hypoxia.
Yong J, Gröger S, von Bremen J, Meyle J, Ruf S
Cells 2022 Jul 30;11(15)
Cells 2022 Jul 30;11(15)
Mitochondrial ROS drive resistance to chemotherapy and immune-killing in hypoxic non-small cell lung cancer.
Salaroglio IC, Belisario DC, Akman M, La Vecchia S, Godel M, Anobile DP, Ortone G, Digiovanni S, Fontana S, Costamagna C, Rubinstein M, Kopecka J, Riganti C
Journal of experimental & clinical cancer research : CR 2022 Aug 11;41(1):243
Journal of experimental & clinical cancer research : CR 2022 Aug 11;41(1):243
LW1497, an Inhibitor of Malate Dehydrogenase, Suppresses TGF-β1-Induced Epithelial-Mesenchymal Transition in Lung Cancer Cells by Downregulating Slug.
Kim HJ, Park MK, Byun HJ, Kim M, Kim B, Yu L, Nguyen TM, Nguyen TH, Do PA, Kim EJ, Kim JH, Enkhtaivan E, Kim KS, Jang JY, Kang GJ, Lee H, Won M, Lee K, Cho J, Lee CH
Antioxidants (Basel, Switzerland) 2021 Oct 24;10(11)
Antioxidants (Basel, Switzerland) 2021 Oct 24;10(11)
Oxidation of Hemoglobin Drives a Proatherogenic Polarization of Macrophages in Human Atherosclerosis.
Potor L, Hendrik Z, Patsalos A, Katona É, Méhes G, Póliska S, Csősz É, Kalló G, Komáromi I, Combi Z, Posta N, Sikura KÉ, Pethő D, Oros M, Vereb G, Tóth C, Gergely P, Nagy L, Balla G, Balla J
Antioxidants & redox signaling 2021 Oct 20;35(12):917-950
Antioxidants & redox signaling 2021 Oct 20;35(12):917-950
Obesity Impairs Embryonic Myogenesis by Enhancing BMP Signaling within the Dermomyotome.
Zhao L, Law NC, Gomez NA, Son J, Gao Y, Liu X, de Avila JM, Zhu MJ, Du M
Advanced science (Weinheim, Baden-Wurttemberg, Germany) 2021 Nov;8(22):e2102157
Advanced science (Weinheim, Baden-Wurttemberg, Germany) 2021 Nov;8(22):e2102157
Human Pluripotent Stem Cell-Derived Neural Progenitor Cells Promote Retinal Ganglion Cell Survival and Axon Recovery in an Optic Nerve Compression Animal Model.
Park M, Kim HM, Shin HA, Lee SH, Hwang DY, Lew H
International journal of molecular sciences 2021 Nov 20;22(22)
International journal of molecular sciences 2021 Nov 20;22(22)
Silicon Oxynitrophosphide Nanoscale Coating Enhances Antioxidant Marker-Induced Angiogenesis During in vivo Cranial Bone-Defect Healing.
do Monte FA, Ahuja N, Awad KR, Pan Z, Young S, Kim HK, Aswath P, Brotto M, Varanasi VG
JBMR plus 2021 Apr;5(4):e10425
JBMR plus 2021 Apr;5(4):e10425
An Important Role for DNMT3A-Mediated DNA Methylation in Cardiomyocyte Metabolism and Contractility.
Madsen A, Höppner G, Krause J, Hirt MN, Laufer SD, Schweizer M, Tan WLW, Mosqueira D, Anene-Nzelu CG, Lim I, Foo RSY, Hansen A, Eschenhagen T, Stenzig J
Circulation 2020 Oct 20;142(16):1562-1578
Circulation 2020 Oct 20;142(16):1562-1578
Maternal erythrocyte ENT1-mediated AMPK activation counteracts placental hypoxia and supports fetal growth.
Sayama S, Song A, Brown BC, Couturier J, Cai X, Xu P, Chen C, Zheng Y, Iriyama T, Sibai B, Longo M, Kellems RE, D'Alessandro A, Xia Y
JCI insight 2020 May 21;5(10)
JCI insight 2020 May 21;5(10)
Oxygen Tension and the VHL-Hif1α Pathway Determine Onset of Neuronal Polarization and Cerebellar Germinal Zone Exit.
Kullmann JA, Trivedi N, Howell D, Laumonnerie C, Nguyen V, Banerjee SS, Stabley DR, Shirinifard A, Rowitch DH, Solecki DJ
Neuron 2020 May 20;106(4):607-623.e5
Neuron 2020 May 20;106(4):607-623.e5
YAP1 and TAZ negatively control bone angiogenesis by limiting hypoxia-inducible factor signaling in endothelial cells.
Sivaraj KK, Dharmalingam B, Mohanakrishnan V, Jeong HW, Kato K, Schröder S, Adams S, Koh GY, Adams RH
eLife 2020 Jan 20;9
eLife 2020 Jan 20;9
Clinicopathological Analysis of HIF-1alpha and TERT on Survival Outcome in Glioblastoma Patients: A Prospective, Single Institution Study.
Potharaju M, Mathavan A, Mangaleswaran B, Patil S, John R, Ghosh S, Kalavakonda C, Ghosh M, Verma RS
Journal of Cancer 2019;10(11):2397-2406
Journal of Cancer 2019;10(11):2397-2406
Enantioselective Synthesis of 8-Hydroxyquinoline Derivative, Q134 as a Hypoxic Adaptation Inducing Agent.
Hackler L Jr, Gyuris M, Huzián O, Alföldi R, Szebeni GJ, Madácsi R, Knapp L, Kanizsai I, Puskás LG
Molecules (Basel, Switzerland) 2019 Nov 23;24(23)
Molecules (Basel, Switzerland) 2019 Nov 23;24(23)
Chloramphenicol Induces Autophagy and Inhibits the Hypoxia Inducible Factor-1 Alpha Pathway in Non-Small Cell Lung Cancer Cells.
Hsu HL, Liao PL, Cheng YW, Huang SH, Wu CH, Li CH, Kang JJ
International journal of molecular sciences 2019 Jan 3;20(1)
International journal of molecular sciences 2019 Jan 3;20(1)
Macrophage-mediated delivery of light activated nitric oxide prodrugs with spatial, temporal and concentration control.
Evans MA, Huang PJ, Iwamoto Y, Ibsen KN, Chan EM, Hitomi Y, Ford PC, Mitragotri S
Chemical science 2018 Apr 21;9(15):3729-3741
Chemical science 2018 Apr 21;9(15):3729-3741
Ginsenoside Rg5 Inhibits Succinate-Associated Lipolysis in Adipose Tissue and Prevents Muscle Insulin Resistance.
Xiao N, Yang LL, Yang YL, Liu LW, Li J, Liu B, Liu K, Qi LW, Li P
Frontiers in pharmacology 2017;8:43
Frontiers in pharmacology 2017;8:43
Hypoxia-Inducible Factor-Dependent Expression of Angiopoietin-Like 4 by Conjunctival Epithelial Cells Promotes the Angiogenic Phenotype of Pterygia.
Meng Q, Qin Y, Deshpande M, Kashiwabuchi F, Rodrigues M, Lu Q, Ren H, Elisseeff JH, Semenza GL, Montaner SV, Sodhi A
Investigative ophthalmology & visual science 2017 Sep 1;58(11):4514-4523
Investigative ophthalmology & visual science 2017 Sep 1;58(11):4514-4523
Hypoxia promotes primitive glycosaminoglycan-rich extracellular matrix composition in developing heart valves.
Amofa D, Hulin A, Nakada Y, Sadek HA, Yutzey KE
American journal of physiology. Heart and circulatory physiology 2017 Dec 1;313(6):H1143-H1154
American journal of physiology. Heart and circulatory physiology 2017 Dec 1;313(6):H1143-H1154
Metformin and resveratrol ameliorate muscle insulin resistance through preventing lipolysis and inflammation in hypoxic adipose tissue.
Zhao W, Li A, Feng X, Hou T, Liu K, Liu B, Zhang N
Cellular signalling 2016 Sep;28(9):1401-1411
Cellular signalling 2016 Sep;28(9):1401-1411
Inhibition of HIF Reduces Bladder Hypertrophy and Improves Bladder Function in Murine Model of Partial Bladder Outlet Obstruction.
Iguchi N, Malykhina AP, Wilcox DT
The Journal of urology 2016 Apr;195(4 Pt 2):1250-6
The Journal of urology 2016 Apr;195(4 Pt 2):1250-6
The Neuroprotective Effect of Coumaric Acid on Spinal Cord Ischemia/Reperfusion Injury in Rats.
Guven M, Sehitoglu MH, Yuksel Y, Tokmak M, Aras AB, Akman T, Golge UH, Karavelioglu E, Bal E, Cosar M
Inflammation 2015 Oct;38(5):1986-95
Inflammation 2015 Oct;38(5):1986-95
Effect of CoCl₂ on fracture repair in a rat model of bone fracture.
Huang J, Liu L, Feng M, An S, Zhou M, Li Z, Qi J, Shen H
Molecular medicine reports 2015 Oct;12(4):5951-6
Molecular medicine reports 2015 Oct;12(4):5951-6
K63 linked ubiquitin chain formation is a signal for HIF1A degradation by Chaperone-Mediated Autophagy.
Ferreira JV, Soares AR, Ramalho JS, Pereira P, Girao H
Scientific reports 2015 May 11;5:10210
Scientific reports 2015 May 11;5:10210
Snail Is a Direct Target of Hypoxia-inducible Factor 1α (HIF1α) in Hypoxia-induced Endothelial to Mesenchymal Transition of Human Coronary Endothelial Cells.
Xu X, Tan X, Tampe B, Sanchez E, Zeisberg M, Zeisberg EM
The Journal of biological chemistry 2015 Jul 3;290(27):16653-64
The Journal of biological chemistry 2015 Jul 3;290(27):16653-64
Wnt signaling directs a metabolic program of glycolysis and angiogenesis in colon cancer.
Pate KT, Stringari C, Sprowl-Tanio S, Wang K, TeSlaa T, Hoverter NP, McQuade MM, Garner C, Digman MA, Teitell MA, Edwards RA, Gratton E, Waterman ML
The EMBO journal 2014 Jul 1;33(13):1454-73
The EMBO journal 2014 Jul 1;33(13):1454-73
HIF-1α-induced HSP70 regulates anabolic responses in articular chondrocytes under hypoxic conditions.
Tsuchida S, Arai Y, Takahashi KA, Kishida T, Terauchi R, Honjo K, Nakagawa S, Inoue H, Ikoma K, Ueshima K, Matsuki T, Mazda O, Kubo T
Journal of orthopaedic research : official publication of the Orthopaedic Research Society 2014 Aug;32(8):975-80
Journal of orthopaedic research : official publication of the Orthopaedic Research Society 2014 Aug;32(8):975-80
Inhibition of mitochondrial complex III blocks neuronal differentiation and maintains embryonic stem cell pluripotency.
Pereira SL, Grãos M, Rodrigues AS, Anjo SI, Carvalho RA, Oliveira PJ, Arenas E, Ramalho-Santos J
PloS one 2013;8(12):e82095
PloS one 2013;8(12):e82095
Response of small heat shock proteins in diabetic rat retina.
Reddy VS, Raghu G, Reddy SS, Pasupulati AK, Suryanarayana P, Reddy GB
Investigative ophthalmology & visual science 2013 Nov 19;54(12):7674-82
Investigative ophthalmology & visual science 2013 Nov 19;54(12):7674-82
Superoxide induced by a high-glucose concentration attenuates production of angiogenic growth factors in hypoxic mouse mesenchymal stem cells.
Ishizuka T, Hinata T, Watanabe Y
The Journal of endocrinology 2011 Feb;208(2):147-59
The Journal of endocrinology 2011 Feb;208(2):147-59
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
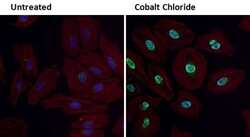
- Experimental details
- Immunofluorescent analysis of HIF-1 alpha (green) in HeLa cells either left untreated (left panel) or treated with 0.1 mM Cobalt Chloride (right panel) for 4 hours. Formalin fixed cells were permeabilized with 0.1% Triton X-100 in TBS for 10 minutes at room temperature and blocked with 1% Blocker BSA (Product # 37525) for 15 minutes at room temperature. Cells were probed with a HIF-1 alpha polyclonal antibody (Product # PA1-16601) at a dilution of 1:100 for at least 1 hour at room temperature, washed with PBS, and incubated with DyLight 488 goat-anti-rabbit IgG secondary antibody (Product # 35552) at a dilution of 1:400 for 30 minutes at room temperature. F-Actin (red) was stained with DyLight-554 Phalloidin (Product # 21834) and nuclei (blue) were stained with Hoechst 33342 dye (Product # 62249). Images were taken on a Thermo Scientific ArrayScan at 20X magnification.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunocytochemistry analysis of HIF1A in RCC4 cells. Samples were incubated in HIF1A polyclonal antibody (Product # PA1-16601). IF-1 Alpha (green). Nuclei (Blue) were counterstained using Hoechst 33258.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunocytochemistry analysis of HIF1A in RCC4 cells. Samples were incubated in HIF1A polyclonal antibody (Product # PA1-16601). IF-1 Alpha (green). Nuclei (Blue) were counterstained using Hoechst 33258.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunofluorescent analysis of HIF-1 alpha (green) in HeLa cells either left untreated (left panel) or treated with 0.1 mM Cobalt Chloride (right panel) for 4 hours. Formalin fixed cells were permeabilized with 0.1% Triton X-100 in TBS for 10 minutes at room temperature and blocked with 1% Blocker BSA (Product # 37525) for 15 minutes at room temperature. Cells were probed with a HIF-1 alpha polyclonal antibody (Product # PA1-16601) at a dilution of 1:100 for at least 1 hour at room temperature, washed with PBS, and incubated with DyLight 488 goat-anti-rabbit IgG secondary antibody (Product # 35552) at a dilution of 1:400 for 30 minutes at room temperature. F-Actin (red) was stained with DyLight-554 Phalloidin (Product # 21834) and nuclei (blue) were stained with Hoechst 33342 dye (Product # 62249). Images were taken on a Thermo Scientific ArrayScan at 20X magnification.
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemical analysis of HIF1A in human placenta, villi. Samples were incubated in HIF1A polyclonal antibody (Product # PA1-16601).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Immunohistochemical analysis of HIF1A in human kidney tumor. Samples were incubated in HIF1A polyclonal antibody (Product # PA1-16601).
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 1 Effects of Rg5 on adipose hypoxia. (A) Adipose hypoxia examination with pimonidazole staining in HFD mice. (B) HIF-1alpha protein expression in adipose tissue of HFD-fed mice. (C,D) HIF-1alpha protein expression in differentiated adipocytes treated with palmitate (PA), 1% O 2 for 8 h. (E,F) Pimonidazole staining in differentiated adipocytes treated with PA for 8 h or 1% O 2 for 4 h. The results were expressed as the mean +- SD of three independent experiments. * p < 0.05 vs . Control (Ctr), # p < 0.05 vs . Blank. TU, Tauroursodeoxycholic Acid; Met, Metformin.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 8 HIF-1alpha protein levels were stabilized by AA treatment. HIF-1alpha protein levels were evaluated by western blot at different time-points upon AA treatment. (A) mESC incubated for 14h with CoCl 2 (300 uM) a known disruptor of prolyl hydroxylase activity, show increased Hif-1alpha protein levels. mESC without CoCl 2 treatment were used as control and different protein volumes were loaded in the gel. (B) Representative blot for E14 cells showing increased Hif-1alpha protein levels downstream to AA treatment. (C) Densiometric evaluation of HIF-1alpha and actin (loading control) bands show that a statistical significant difference exists between control and treated conditions at 2 and 4 hours for both E14 and R1 mESC. Data shown as protein level fold increase for each one of the AA conditions relative to the respective control. Data were normal and evaluated by one sample T Student tests against a theoretical value of 1. Error bars = SEM and statistical significance is considered when * p
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 Accumulation of HIF-1alpha in cultured CjECs results in the secretion of angiogenic mediators. (A) Immunoblot for HIF-1alpha in ihCjECs exposed to hypoxia (1% O 2 ) or a HIF inducer (100 muM DFO or 300 muM DMOG), in normoxia (20% O 2 ) for 4 hours. One hundred nanomolar digoxin was used to inhibit HIF-1alpha accumulation. (B) Immunoblot for HIF-1alpha in prCjECs exposed to 1% O 2 or 300 muM DMOG for 4 hours. (C) H&E stain of stratified prCjECs grown on vitrigel. (D) Immunoblot for HIF-1alpha in stratified prCjECs grown on vitrigel exposed to 1% O 2 or 300 muM DMOG for 4 hours. (E-H) EC tubule formation by HMVECs treated with conditioned media from ihCjECs exposed to 1% O 2 (E, F) or 100 muM DFO or 300 muM DMOG (G, H), in the absence (E, G) or presence (F, H) of 100 nM digoxin, compared to media conditioned by cells exposed to 20% O 2 for 24 hours. Ten percent FBS was used as a positive control.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig 10 Immunofluorescence staining (Alexa Fluor 594) for Ang-1 and HIF-1alpha, 15 days after implantation. Scale bar 100 mum. ( A ) SiONPx2. ( B ) Uncoated ""bare"" implant. ( C ) Percentage of area occupied by Ang-1. Statistical difference, *** p < 0.001. ( D ) SiONPx2. ( E ) Uncoated ""bare"" implant. ( F ) Percentage of area occupied by HIF-1alpha. Statistical difference, * p < 0.05. Ang-1 = Angiopoietin-1; HIF-1alpha = hypoxia inducible factor-1 alpha; SiONP = silicon oxynitrophosphide.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5 LW1497 suppressed lung cancer progression in a lung cancer orthotopic mouse model. ( A ) In vivo experimental protocol of A549 cells in NOD/SCID mouse using LW1497 drug as described in Methods section. ( B ) Hematoxylin and eosin staining of the lungs in LW1497- or vehicle-treated NOD/SCID mice injected with A549 cells. Lungs were analyzed by hematoxylin and eosin staining. ( C ) Comparison of tumor areas: mice treated with LW1497 versus mice treated with vehicle. Tumor areas were measured by intravital microscopic analysis (Axiotech Vario microscope, Zeiss, Germany). * p < 0.05, ** p < 0.01 compared with the control group. ( D ) Immunohistochemical analysis of Ki67, HIF-1a, Slug, and EMT markers including Vimentin (Vim), E-cadherin (E-cad), and N-cadherin (N-cad) in lung tumors in LW1497 (10, 30 mg/kg)- or vehicle-treated mice orthotopically injected with A549 cells into the lung.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 2 Human NPCs have the function of recovering damaged R28 cells. R28 cells were cultured with NPCs or hPSCs 3 h prior to CoCl 2 treatment. Then, the R28 cells were treated with CoCl 2 (300 muM). ( A ) Viability assays performed after 24 h. Data are expressed as percentage (mean +- SEM) of viable cells compared to those in the control group. Significantly different values between groups are indicated by different letters ( p < 0.05). ( B ) Apoptosis-related protein expressions were also determined. ( C ) Western blot analyses of target protein expression levels, using R28 lysates with CoCl 2 . The quantified values of target protein expression are presented (bottom panel) (* p < 0.05 vs. the control; # p < 0.05, ## p < 0.01 vs. CoCl 2; + p < 0.05 vs. PSCs).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- FIG. 14. PI3K/HIF-1alpha/VEGF pathway in macrophages exposed to ferryl hemoglobin. (A) Human macrophages were treated with 10 mu M oxyHb or ferrylHb for 2 h. The expression of PI3K and phopsho-PI3K protein was assessed by Western blot ( n = 3). (B) Human macrophages were treated with 10 mu M heme, oxyHb, metHb, or ferrylHb for 16 h. The expression of HIF-1alpha was assessed by Western blot ( n = 5). (C) Human macrophages were treated with 10 mu M oxyHb or ferrylHb for 24 h. The secreted VEGF-A was measured by the ELISA-kit. * p < 0.5; ** p < 0.01. ELISA, enzyme-linked immunosorbent assay; HIF-1alpha, hypoxia-inducible factor 1-alpha; PI3K, phosphoinositide 3-kinase; VEGF, vascular endothelial growth factor.
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- CoCl 2 -induced hypoxia differentially affects HIF-1alpha expression in cementoblasts. ( A , B ) RT-qPCR quantification of HIF-1alpha gene expression in OCCM-30 cells cultured under normoxia or hypoxia (100, 400 muM) conditions for indicated timepoints. The relative mRNA expression of HIF-1alpha was obtained through normalizing to internal PPIB. Each dot, triangle and square symbol represent a relative PD-L1 expression value. ( C , D ) HIF-1alpha protein levels were determined by Western blot. Internal beta-actin levels served as loading control. Quantification indicated the enhanced HIF-1alpha expression in 8 to 24 h in the 400 muM hypoxia treated group relative to the control group. Results are reported as fold change and relative the control group as 1. ( E ) Confocal microscopy analysis of subcellular localization of HIF-1alpha (red) in OCCM-30 cells cultured under normoxia or hypoxic conditions for 24 h at the same initial cell density. White arrows show its cellular localization. Nuclei are stained with DAPI (blue). Individual and merged images of HIF-1alpha and DAPI are shown. Scale bar: 100 mum. The blots and photomicrographs represent three independent experiments with SD. Statistically significant differences (indicated by asterisks) are indicated as follows (ns, no significant difference; * p < 0.05; ** p < 0.005).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 3 A major role of HIF-1alpha inhibition on HIF-1alpha expression under hypoxia. Cells were preincubated with different concentrations of HIF-1alpha inhibitor (IDF11774) for 2 h and then incubated in hypoxic conditions for 24 h, respectively. ( A ) Western blot analysis of the expression levels of HIF-1alpha in the presence or absence of IDF-11774 (0, 5, 10, 20 nM). HIF-1alpha accumulation was reduced by the increased concentration of IDF-11774. Internal beta-actin levels serve as loading control. ( B ) Quantification was performed to detected protein changes of HIF-1alpha, and it was demonstrated that its expression is significantly inhibited by the highest concentration of IDF-11774 (20 nM). ( C , D ) Representative IF staining showing the distribution changes of HIF-1alpha in hypoxia-treated OCCM-30 cells with the treatment of the pharmacological signaling inhibitor IDF-11774 (20 nM). Yellow arrows indicate the expression of HIF-1alpha was effectively inhibited by the HIF-1alpha inhibitor IDF-11774. ( E ) Verification of the efficacy of HIF-1alpha CRISPR/Cas9 system. Knockdown of HIF-1alpha gene expression by CRISPR/Cas9 decreased the hypoxia-induced gene expression of HIF-1alpha in OCCM-30 cells. The blots and photomicrographs represent three independent experiments with SD. Statistically significant differences (indicated by asterisks) are indicated as follows (ns, no significant difference; * p < 0.05; ** p < 0.005).
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5. Yap1 and Taz inhibit HIF-1alpha-controlled gene expression. ( A ) Increased expression of the HIF1alpha target genes VEGFA and ANGPTL4 as well as Yap1/Taz target genes CTGF and CYR61 under hypoxic condition (1% O 2 ) compared to normoxia (21% O 2 ) (n = 4; data are presented as mean +-sem, P values, two-tailed unpaired t-test ). ( B ) Confocal image of HUVEC showing nuclear accumulation of Yap1/Taz in 1% O 2 relative to 21% O 2 . Nuclei, DAPI (blue). ( C ) Increased expression of the Yap1/Taz target genes CTGF, CYR61 and the HIF1alpha target VEGFA in HUVECs in 1% O 2 relative to 21% O 2 . CTGF and CYR61 expression under both conditions is significantly reduced in siYAP1/TAZ -transfected (Y/T) HUVECs. VEGFA expression significantly increased in Y/T cells in both conditions (n = 4; data are presented as mean +-sem, P values, two-tailed unpaired t-test ). ( D ) Western blots showing immunoprecipitation (IP) of Yap1/Taz followed by immunoblotting of HIF1alpha and Yap1/Taz. IgG is used as negative control in IP. Lysates are shown as loading control. ( E ) Increased VEGFA , ANGPTL4 , IGFBP2 expression in siYAP1/TAZ -treated HUVECs is normalized by siHIF1A transfection, whereas baseline VEGFA , ANGPTL4 and IGFBP2 is not altered by HIF1A knockdown alone (n = 3-4; data are presented as mean +-sem, P values, two-tailed unpaired t-test ). ( F ) Enrichment of VEGFA promoter sequences after chromatin immunoprecipitation with HIF1alpha antibodies compared to IgG antibodies under
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6 Chloramphenicol interrupted protein interaction between SENP-1 and HIF-1alpha and promoted HIF-1alpha degradation via the autophagy pathway. ( A ) Representative images of co-immunoprecipitation assays performed with HIF-1alpha-overexpressing H1299 cells in the presence or absence of chloramphenicol. Whole cell lysates was immunoprecipitated with anti-HIF-1alpha (left) or anti-SENP-1 (right) antibody, followed by immunoblotting analysis. A protein complex of HIF-1alpha/SENP-1 was found in the control treatment. Chloramphenicol treatment not only reduced HIF-1alpha levels in whole lysate, but also inhibited HIF-1alpha/SENP-1 interaction. ( B ) Whole lysate of HIF-1alpha-overexpressing H1299 was prepared and aliquoted (1 mg protein/vial). Before reaction with the captured antibody, one vial of sample was incubated with chloramphenicol (100 mug/mL) at 4 degC for 1 h. Another vial, without chloramphenicol treatment, was used as a control. Then, the co-immunoprecipitation was performed as described previously. The input data showed that neither HIF-1 alpha nor SENP-1 protein level was changed in whole lysates, whatever the presence or absence of chloramphenicol treatment. The HIF-1alpha/SENP-1 protein complex, which was clearly found in lysate without the chloramphenicol treatment, was diminished in lysate with the chloramphenicol treatment. This data provides substantial evidence to support the interaction between HIF-1alpha and SENP-1 was damaged by chloramphenicol. (
 Explore
Explore Validate
Validate Learn
Learn