Antibody data
- Antibody Data
- Antigen structure
- References [19]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [13]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 11-0389-42 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD38 Monoclonal Antibody (HIT2), FITC, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Description
- Description: The HIT2 monoclonal antibody reacts with the human CD38 molecule, an approximately 45 kDa type II transmembrane protein. CD38 is expressed by thymocytes, peripheral B cells including plasma cells, activated T cells, and monocytes. CD38 is a counter-receptor for CD31 and has both cyclase and hydrolase enzymatic activity. Applications Reported: The HIT2 antibody has been reported for use in flow cytometric analysis. Applications Tested: The HIT2 antibody has been pre-titrated and tested by flow cytometric analysis of normal human peripheral blood cells. This can be used at 5 µL (1 µg) per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. Excitation: 488 nm; Emission: 520 nm; Laser: Blue Laser. Filtration: 0.2 µm post-manufacturing filtered.
- Reactivity
- Human
- Host
- Mouse
- Conjugate
- Green dye
- Isotype
- IgG
- Antibody clone number
- HIT2
- Vial size
- 100 Tests
- Concentration
- 5 µL/Test
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references Immunometabolic interference between cancer and COVID-19.
Novel myeloma patient-derived xenograft models unveil the potency of anlotinib to overcome bortezomib resistance.
B cell-intrinsic changes with age do not impact antibody-secreting cell formation but delay B cell participation in the germinal centre reaction.
Enhanced CXCR4 Expression of Human CD8(Low) T Lymphocytes Is Driven by S1P(4).
SARS-CoV-2 infection paralyzes cytotoxic and metabolic functions of the immune cells.
Kaposi's sarcoma-associated herpesvirus latency-associated nuclear antigen dysregulates expression of MCL-1 by targeting FBW7.
Highly efficient CRISPR-Cas9-mediated gene knockout in primary human B cells for functional genetic studies of Epstein-Barr virus infection.
Synthesis of site-specific antibody-drug conjugates by ADP-ribosyl cyclases.
Chronic myelomonocytic leukaemia stem cell transcriptomes anticipate disease morphology and outcome.
IFNγ induces epigenetic programming of human T-bet(hi) B cells and promotes TLR7/8 and IL-21 induced differentiation.
Abnormal phenotypic features of IgM+B cell subsets in patients with chronic hepatitis C virus infection.
Heterogeneous leukemia stem cells in myeloid blast phase chronic myeloid leukemia.
The upregulation of LAG-3 on T cells defines a subpopulation with functional exhaustion and correlates with disease progression in HIV-infected subjects.
Generation of functional, antigen-specific CD8+ human T cells from cord blood stem cells using exogenous Notch and tetramer-TCR signaling.
Incomplete normalization of regulatory t-cell frequency in the gut mucosa of Colombian HIV-infected patients receiving long-term antiretroviral treatment.
CD4⁺CD73⁺ T cells are associated with lower T-cell activation and C reactive protein levels and are depleted in HIV-1 infection regardless of viral suppression.
Targeting CDK1 promotes FLT3-activated acute myeloid leukemia differentiation through C/EBPα.
Generation of bivalent chromatin domains during cell fate decisions.
CD38 binding to human myeloid cells is mediated by mouse and human CD31.
Consonni FM, Durante B, Manfredi M, Bleve A, Pandolfo C, Garlatti V, Vanella VV, Marengo E, Barberis E, Bottazzi B, Bombace S, My I, Condorelli G, Torri V, Sica A
Frontiers in immunology 2023;14:1168455
Frontiers in immunology 2023;14:1168455
Novel myeloma patient-derived xenograft models unveil the potency of anlotinib to overcome bortezomib resistance.
Yue Y, Cao Y, Mao X, Wang F, Fan P, Qian L, Guo S, Li F, Guo Y, Chen T, Lin Y, Dong W, Liu Y, Huang Y, Gu W
Frontiers in oncology 2022;12:894279
Frontiers in oncology 2022;12:894279
B cell-intrinsic changes with age do not impact antibody-secreting cell formation but delay B cell participation in the germinal centre reaction.
Lee JL, Fra-Bido SC, Burton AR, Innocentin S, Hill DL, Linterman MA
Aging cell 2022 Sep;21(9):e13692
Aging cell 2022 Sep;21(9):e13692
Enhanced CXCR4 Expression of Human CD8(Low) T Lymphocytes Is Driven by S1P(4).
Burkard T, Dreis C, Herrero San Juan M, Huhn M, Weigert A, Pfeilschifter JM, Radeke HH
Frontiers in immunology 2021;12:668884
Frontiers in immunology 2021;12:668884
SARS-CoV-2 infection paralyzes cytotoxic and metabolic functions of the immune cells.
Singh Y, Trautwein C, Fendel R, Krickeberg N, Berezhnoy G, Bissinger R, Ossowski S, Salker MS, Casadei N, Riess O, Deutsche COVID-19 OMICS Initiate (DeCOI)
Heliyon 2021 Jun;7(6):e07147
Heliyon 2021 Jun;7(6):e07147
Kaposi's sarcoma-associated herpesvirus latency-associated nuclear antigen dysregulates expression of MCL-1 by targeting FBW7.
Kim YJ, Kim Y, Kumar A, Kim CW, Toth Z, Cho NH, Lee HR
PLoS pathogens 2021 Jan;17(1):e1009179
PLoS pathogens 2021 Jan;17(1):e1009179
Highly efficient CRISPR-Cas9-mediated gene knockout in primary human B cells for functional genetic studies of Epstein-Barr virus infection.
Akidil E, Albanese M, Buschle A, Ruhle A, Pich D, Keppler OT, Hammerschmidt W
PLoS pathogens 2021 Apr;17(4):e1009117
PLoS pathogens 2021 Apr;17(4):e1009117
Synthesis of site-specific antibody-drug conjugates by ADP-ribosyl cyclases.
Dai Z, Zhang XN, Nasertorabi F, Cheng Q, Li J, Katz BB, Smbatyan G, Pei H, Louie SG, Lenz HJ, Stevens RC, Zhang Y
Science advances 2020 Jun;6(23):eaba6752
Science advances 2020 Jun;6(23):eaba6752
Chronic myelomonocytic leukaemia stem cell transcriptomes anticipate disease morphology and outcome.
Wiseman DH, Baker SM, Dongre AV, Gurashi K, Storer JA, Somervaille TC, Batta K
EBioMedicine 2020 Aug;58:102904
EBioMedicine 2020 Aug;58:102904
IFNγ induces epigenetic programming of human T-bet(hi) B cells and promotes TLR7/8 and IL-21 induced differentiation.
Zumaquero E, Stone SL, Scharer CD, Jenks SA, Nellore A, Mousseau B, Rosal-Vela A, Botta D, Bradley JE, Wojciechowski W, Ptacek T, Danila MI, Edberg JC, Bridges SL Jr, Kimberly RP, Chatham WW, Schoeb TR, Rosenberg AF, Boss JM, Sanz I, Lund FE
eLife 2019 May 15;8
eLife 2019 May 15;8
Abnormal phenotypic features of IgM+B cell subsets in patients with chronic hepatitis C virus infection.
Kong F, Feng B, Zhang H, Rao H, Wang J, Cong X, Wei L
Experimental and therapeutic medicine 2017 Aug;14(2):1846-1852
Experimental and therapeutic medicine 2017 Aug;14(2):1846-1852
Heterogeneous leukemia stem cells in myeloid blast phase chronic myeloid leukemia.
Kinstrie R, Karamitros D, Goardon N, Morrison H, Hamblin M, Robinson L, Clark RE, Copland M, Vyas P
Blood advances 2016 Dec 27;1(3):160-169
Blood advances 2016 Dec 27;1(3):160-169
The upregulation of LAG-3 on T cells defines a subpopulation with functional exhaustion and correlates with disease progression in HIV-infected subjects.
Tian X, Zhang A, Qiu C, Wang W, Yang Y, Qiu C, Liu A, Zhu L, Yuan S, Hu H, Wang W, Wei Q, Zhang X, Xu J
Journal of immunology (Baltimore, Md. : 1950) 2015 Apr 15;194(8):3873-82
Journal of immunology (Baltimore, Md. : 1950) 2015 Apr 15;194(8):3873-82
Generation of functional, antigen-specific CD8+ human T cells from cord blood stem cells using exogenous Notch and tetramer-TCR signaling.
Fernandez I, Ooi TP, Roy K
Stem cells (Dayton, Ohio) 2014 Jan;32(1):93-104
Stem cells (Dayton, Ohio) 2014 Jan;32(1):93-104
Incomplete normalization of regulatory t-cell frequency in the gut mucosa of Colombian HIV-infected patients receiving long-term antiretroviral treatment.
Rueda CM, Velilla PA, Chougnet CA, Rugeles MT
PloS one 2013;8(8):e71062
PloS one 2013;8(8):e71062
CD4⁺CD73⁺ T cells are associated with lower T-cell activation and C reactive protein levels and are depleted in HIV-1 infection regardless of viral suppression.
Schuler PJ, Macatangay BJ, Saze Z, Jackson EK, Riddler SA, Buchanan WG, Hilldorfer BB, Mellors JW, Whiteside TL, Rinaldo CR
AIDS (London, England) 2013 Jun 19;27(10):1545-55
AIDS (London, England) 2013 Jun 19;27(10):1545-55
Targeting CDK1 promotes FLT3-activated acute myeloid leukemia differentiation through C/EBPα.
Radomska HS, Alberich-Jordà M, Will B, Gonzalez D, Delwel R, Tenen DG
The Journal of clinical investigation 2012 Aug;122(8):2955-66
The Journal of clinical investigation 2012 Aug;122(8):2955-66
Generation of bivalent chromatin domains during cell fate decisions.
De Gobbi M, Garrick D, Lynch M, Vernimmen D, Hughes JR, Goardon N, Luc S, Lower KM, Sloane-Stanley JA, Pina C, Soneji S, Renella R, Enver T, Taylor S, Jacobsen SE, Vyas P, Gibbons RJ, Higgs DR
Epigenetics & chromatin 2011 Jun 6;4(1):9
Epigenetics & chromatin 2011 Jun 6;4(1):9
CD38 binding to human myeloid cells is mediated by mouse and human CD31.
Horenstein AL, Stockinger H, Imhof BA, Malavasi F
The Biochemical journal 1998 Mar 15;330 ( Pt 3):1129-35
The Biochemical journal 1998 Mar 15;330 ( Pt 3):1129-35
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Staining of normal human peripheral blood cells with Mouse IgG1 K Isotype Control FITC (Product # 11-4714-42) (blue histogram) or Anti-Human CD38 FITC (purple histogram). Cells in the lymphocyte gate (left) and monocyte gate (right) were used for analysis.
- Conjugate
- Green dye
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4. IFNgamma is required for development of T-bet hi B DN cells and regulates ASC formation and recovery. ( a ) Ingenuity Pathway Analysis (IPA) to identify predicted upstream direct and indirect regulators of the HD B DN Be1 transcriptome. IPA performed using the 427 DEG (B DN Be1 over B DN Be2; FDR < 0.05) identified in the RNA-seq analysis described in Figure 3b . The predicted activation state (z-score of B DN Be1 over B DN Be2) of each regulator/signaling pathway is shown as bar color (orange, activated; blue, inhibited) with predicted upstream regulators sorted in order of significance (overlap P value). Regulators with an overlap P -value
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5. Temporally distinct regulation of T-bet hi IRF4 int pre-ASC and ASC development by IFNgamma, R848 and IL-21. Cartoon ( a ) depicting stimulation of CTV-labeled HD B N cells for 3 days with anti-Ig, R848, IL-21 and IFNgamma (Step 1). Cells were washed and re-cultured for 3 days with R848, IFNgamma, and IL-21 (Step 2, +,+ condition) or individual stimuli were included in Step 1 only (+,- condition) or in Step 2 only (-,+ condition). Cells from day 6 cultures containing IFNgamma ( b-e ), R848 ( f-i ) or IL-21 ( j-m ) in Step 1, Step 2 or both steps were analyzed to determine ASC frequencies ( b, f, j ), ASC recovery ( c, g, k ), cell division ( d, h, l ) and total cell recovery ( e, i, m ). Summary of data ( n ) showing that ASC development and recovery from T-bet hi IRF4 int B DN pre-ASCs requires early IFNgamma, R848 and BCR ''priming'' signals and late R848 and IL-21 proliferation and differentiation signals. See Figure 5-figure supplement 1 for representative flow cytometry plots from each culture showing T-bet hi IRF4 int B DN cells on day 3, CD38 hi CD27 + ASCs on day 6 and CTV dilution on day 6. Data are representative of >=3 experiments. The percentage of cells in each division, the frequency of ASCs and cell recovery (total and ASCs) are shown as the mean +-SD of cultures containing purified B N cells from 3 independent healthy donors. All statistical analyses were performed using one-way ANOVA with Tukey''s multiple comparison test. P values *
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
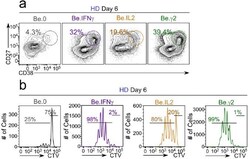
- Figure 6-figure supplement 2. Flow cytometric characterization of B cells activated during the early priming phase in the presence or absence of IFNgamma and IL-2. Day 6 Be.0, Be.IL2, Be.IFNgamma and Be.gamma2 cells were generated as described in Figure 6e . Representative flow cytometry plots from day 6 Be.0, Be.IFNgamma, Be.IL2 and Be.gamma2 cultures showing CD38 hi CD27 + ASCs ( a ) and CTV dilution ( b ).
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6. IFNgamma cooperates with R848, IL-2 and IL-21 to promote development and recovery of ASCs. ( a-d ) IFNgamma synergizes with subthreshold amounts of TLR7/8 ligand to induce proliferation and differentiation of B N cells. CTV-labeled HD B N cells were activated for 3 days (Step 1) with anti-Ig, IL-2, and increasing concentrations of R848 (as indicated) in the presence or absence of IFNgamma (10 ng/ml). Cells were washed and re-cultured for 3 additional days (Step 2) with IL-21 and the same concentration of R848 that was used in Step 1. B cell division was measured on day 6 in cultures that were activated with IFNgamma (green circles) or without IFNgamma (orange circles) in the presence of no R848 (0 mug/ml, ( a ), high dose R848 (10 mug/ml, ( b ) or low dose R848 (0.1 mug/ml, ( c ). The frequency of CD38 hi CD27 + ASCs ( d ) on day 6 is shown. ( e-i ) IFNgamma cooperates with IL-2 to promote ASC development and recovery. Cartoon ( e ) depicting CTV-labeled HD B N cells activated for 3 days (Step 1) with anti-Ig and R848 alone (Be.0); with anti-Ig +R848+IFNgamma (Be.IFNgamma); with anti-Ig +R848+IL-2 (Be.IL2); or with anti-Ig +R848+IFNgamma+IL-2 (Be.gamma2). Cells were then washed and recultured for an additional 3 days (Step 2) with R848 and IL-21. The percentage of cells that have undergone cell division ( f ), the total cell recovery ( g ), the ASC frequencies ( h ) and total ASCs recovered ( i ) from each day 6 culture are shown. ( j-k ) Early IFNgamma signals regu
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
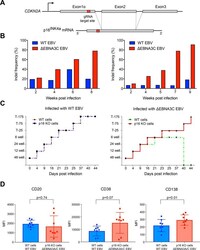
- Fig 4 p16 INK4a is a functional barrier to EBV driven proliferation of lymphoblastoid cells. (A) Blueprint of the primary transcript and the spliced mRNA with the three exons of CDKN2A on chromosome 9 encoding the p16 INK4a protein. The target site of the RNP complex within the 1st exon (exon1alpha) (chr9:21,974,678-21,974,827) is shown. (B) Study of the biological effect of the CDKN2A knockout in a time course experiment. WT and p16 KO cells were mixed such that the fraction of the latter was in the order of 10 to 20%, when the cells were infected with WT or DeltaEBNA3C EBV strains. The knockout status of the CDKN2A gene was studied by next generation sequencing to analyze the CD46 locus of the mixed cell populations over time. The fraction of cells with a disabled CDKN2A gene increased in cells infected with DeltaEBNA3C EBV exceeding 80% after eight weeks, whereas the knockout status of CDKN2A in the population of cells infected with WT EBV did not show a clear trend. Results from two biological replicates are shown, additional replicates can be found in S4A Fig . (C) Cell numbers of four different B cell populations were plotted as a function of days post nucleofection (x-axis) versus the format of the cell culture vessel (y-axis) starting with a single well in a 48-well cluster plate. 2x10 6 B cells with an intact CDKN2A locus (WT cells) or cells with an edited CDKN2A gene (p16 KO cells) were infected with wild-type (WT) EBV (left panel) or DeltaEBNA3C EBV (right panel).
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 7 Increased exhausted CD8 + T cells in convalescent patients. A. Expression of activation marker CD38 on CD8 + T cells (upper FACS panel). One exemplary dot plot is shown per study group. The bar diagram (lower panel) shows that CD38 expression was significantly higher on CD8 + T cells in convalescent COVID-19 + patients compared with HC. *P-value
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
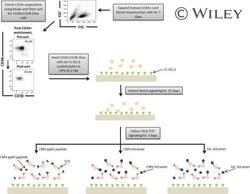
- A schematic overview of our experimental design to generate antigen-specific CD8+ T cells. CD34+ CB cells were expanded for 7 days and CD34+CD38-/low cells were isolated. After 7 days of CD34+ cell expansion, CD34+ cells were enriched using magnetic bead separation. The enriched cells were stained with anti-CD34 and anti-CD38 antibodies to isolate the CD34+CD38-/low HSC population. 81.0% of FSC versus SSC gated cells were CD34+CD38-/low. Gate was determined by the isotype staining control. Data are representative of at least six independent experiments. To induce Notch signaling and early T cell differentiation, CD34+CD38-/low cells were seeded onto DLL1-coated nontissue culture-treated plates. HLA-TCR signaling to direct antigen-specific differentiation was induced using CMVpp65 peptide or epitope-loaded HLA-A*0201 class I tetramers or GIL epitope-loaded HLA-A*0201 class I tetramers. Abbreviations: CM, conditioned medium; CMV, cytomegalovirus; FSC, Forward Scatter; GIL, Influenza M1 virus; HLA, Human leukocyte antigen; SSC, Side Scatter; TCR, T cell receptor.
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
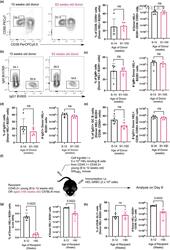
- 7 FIGURE B cells from aged donor mice do not have defects in participating in the GC 10 days after immunization in young recipient mice and fewer B cells from young donor mice are recovered when transferred into aged recipient mice. (a) Representative flow cytometric plots of CD38 - CD95 + GC B cells gated on donor cells from 10-week-old or 93-week-old donor mice in recipient spleens on day 10 post-transfer and immunisation. Numbers adjacent to gates indicate percentage of donor HEL + B220 + cells. Percentage and number of donor-derived GC B cells (Donor HEL + B220 + CD38 - CD95 + ) are plotted on the graphs on the right. (b) Representative flow cytometric plots of IgM + and IgG1 + donor HEL + B cells. Numbers adjacent to gates indicate percentage of donor HEL + B220 + cells. (c, d) Percentage and number of (c) IgM + and (d) IgG1 + B cells derived from donor cells from young or aged mice in recipient spleens 10 days post-transfer and immunisation. (e) Graph showing the percentage and number of IgG1 + GC B cells out of total Donor HEL + B220 + CD38 - CD95 + cells. (f) Schematic diagram of adoptive transfer of B cells from young SW HEL mice into young (8-12 weeks old) or aged (>90 weeks old) mice in which B cells response was analysed 6 days post-transfer and immunisation. (g) Percentage and number of donor HEL + B220 + cells in spleens of young or aged recipient mice 6 days post-transfer and immunisation. (h) Percentage and number of donor-derived GC B cells (Donor HEL + B220
- Conjugate
- Green dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
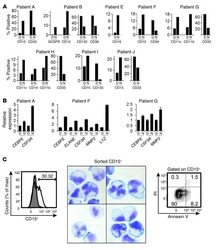
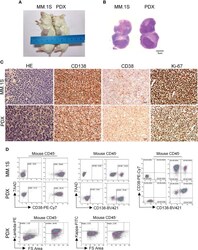
- Morphological and phenotypic features of tumor cells from newly established myeloma PDX. MM.1S cells and mononuclear cells isolated from BM and pleural effusion samples of MM patient were inoculated subcutaneously in NDG mice. When tumors reached 6-8 mm in diameter, tumors were removed and prepared for HE staining, IHC staining, and single-cell suspensions. (A) Representative image of myeloma MM.1S and PDX models. (B) The morphology of tumor cells from myeloma MM.1S and PDX models by Giemsa staining. Scale bar: 5 mum. (C) Representative IHC stainings of tumor tissues. Scale bar: 100 mum. (D) The single-cell suspensions were analyzed by flow cytometry. The doublet or aggregated events were gated out according to side scatter area (SSC-A) and side scatter width (SSC-W). 7-AAD staining was used to gate out dead cells. The expression levels of CD138, CD38, kappa, and lambda were analyzed in mouse CD45 - cells. Myeloma cell line MM.1S was used as a positive myeloma cell control. The left panels of flow charts were isotype controls. PDX, patient-derived xenograft; HE, hematoxylin-eosin; IHC, immunohistochemical.
- Conjugate
- Green dye
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry