Antibody data
- Antibody Data
- Antigen structure
- References [18]
- Comments [0]
- Validations
- Flow cytometry [1]
- Other assay [13]
Submit
Validation data
Reference
Comment
Report error
- Product number
- 56-0389-41 - Provider product page

- Provider
- Invitrogen Antibodies
- Product name
- CD38 Monoclonal Antibody (HIT2), Alexa Fluor™ 700, eBioscience™
- Antibody type
- Monoclonal
- Antigen
- Other
- Reactivity
- Human
- Host
- Mouse
- Conjugate
- Near infrared dye
- Isotype
- IgG
- Antibody clone number
- HIT2
- Vial size
- 25 Tests
- Concentration
- 5 µL/Test
- Storage
- 4° C, store in dark, DO NOT FREEZE!
Submitted references SARS-CoV-2 infection paralyzes cytotoxic and metabolic functions of the immune cells.
Kaposi's sarcoma-associated herpesvirus latency-associated nuclear antigen dysregulates expression of MCL-1 by targeting FBW7.
Highly efficient CRISPR-Cas9-mediated gene knockout in primary human B cells for functional genetic studies of Epstein-Barr virus infection.
IFNγ induces epigenetic programming of human T-bet(hi) B cells and promotes TLR7/8 and IL-21 induced differentiation.
Relative safety of various spermatogenic stem cell purification methods for application in spermatogenic stem cell transplantation.
Intrathecal B Cells in MS Have Significantly Greater Lymphangiogenic Potential Compared to B Cells Derived From Non-MS Subjects.
ATP Release from Chemotherapy-Treated Dying Leukemia Cells Elicits an Immune Suppressive Effect by Increasing Regulatory T Cells and Tolerogenic Dendritic Cells.
Abnormal phenotypic features of IgM+B cell subsets in patients with chronic hepatitis C virus infection.
Antithymocyte Globulin at Clinically Relevant Concentrations Kills Leukemic Blasts.
Single-cell profiling of human megakaryocyte-erythroid progenitors identifies distinct megakaryocyte and erythroid differentiation pathways.
A novel autosomal recessive TERT T1129P mutation in a dyskeratosis congenita family leads to cellular senescence and loss of CD34+ hematopoietic stem cells not reversible by mTOR-inhibition.
Generation of functional, antigen-specific CD8+ human T cells from cord blood stem cells using exogenous Notch and tetramer-TCR signaling.
Inhibitory Receptor Expression Depends More Dominantly on Differentiation and Activation than "Exhaustion" of Human CD8 T Cells.
Development of Norwalk virus-specific monoclonal antibodies with therapeutic potential for the treatment of Norwalk virus gastroenteritis.
Hypermutation of the inactive X chromosome is a frequent event in cancer.
Targeting CDK1 promotes FLT3-activated acute myeloid leukemia differentiation through C/EBPα.
Multiparametric flow cytometry for identification and fluorescence activated cell sorting of five distinct B-cell subpopulations in normal tonsil tissue.
CD38 binding to human myeloid cells is mediated by mouse and human CD31.
Singh Y, Trautwein C, Fendel R, Krickeberg N, Berezhnoy G, Bissinger R, Ossowski S, Salker MS, Casadei N, Riess O, Deutsche COVID-19 OMICS Initiate (DeCOI)
Heliyon 2021 Jun;7(6):e07147
Heliyon 2021 Jun;7(6):e07147
Kaposi's sarcoma-associated herpesvirus latency-associated nuclear antigen dysregulates expression of MCL-1 by targeting FBW7.
Kim YJ, Kim Y, Kumar A, Kim CW, Toth Z, Cho NH, Lee HR
PLoS pathogens 2021 Jan;17(1):e1009179
PLoS pathogens 2021 Jan;17(1):e1009179
Highly efficient CRISPR-Cas9-mediated gene knockout in primary human B cells for functional genetic studies of Epstein-Barr virus infection.
Akidil E, Albanese M, Buschle A, Ruhle A, Pich D, Keppler OT, Hammerschmidt W
PLoS pathogens 2021 Apr;17(4):e1009117
PLoS pathogens 2021 Apr;17(4):e1009117
IFNγ induces epigenetic programming of human T-bet(hi) B cells and promotes TLR7/8 and IL-21 induced differentiation.
Zumaquero E, Stone SL, Scharer CD, Jenks SA, Nellore A, Mousseau B, Rosal-Vela A, Botta D, Bradley JE, Wojciechowski W, Ptacek T, Danila MI, Edberg JC, Bridges SL Jr, Kimberly RP, Chatham WW, Schoeb TR, Rosenberg AF, Boss JM, Sanz I, Lund FE
eLife 2019 May 15;8
eLife 2019 May 15;8
Relative safety of various spermatogenic stem cell purification methods for application in spermatogenic stem cell transplantation.
Tian J, Ma K, Pei CB, Zhang SH, Li X, Zhou Y, Yan B, Wang HY, Ma LH
Stem cell research & therapy 2019 Dec 16;10(1):382
Stem cell research & therapy 2019 Dec 16;10(1):382
Intrathecal B Cells in MS Have Significantly Greater Lymphangiogenic Potential Compared to B Cells Derived From Non-MS Subjects.
Stein J, Xu Q, Jackson KC, Romm E, Wuest SC, Kosa P, Wu T, Bielekova B
Frontiers in neurology 2018;9:554
Frontiers in neurology 2018;9:554
ATP Release from Chemotherapy-Treated Dying Leukemia Cells Elicits an Immune Suppressive Effect by Increasing Regulatory T Cells and Tolerogenic Dendritic Cells.
Lecciso M, Ocadlikova D, Sangaletti S, Trabanelli S, De Marchi E, Orioli E, Pegoraro A, Portararo P, Jandus C, Bontadini A, Redavid A, Salvestrini V, Romero P, Colombo MP, Di Virgilio F, Cavo M, Adinolfi E, Curti A
Frontiers in immunology 2017;8:1918
Frontiers in immunology 2017;8:1918
Abnormal phenotypic features of IgM+B cell subsets in patients with chronic hepatitis C virus infection.
Kong F, Feng B, Zhang H, Rao H, Wang J, Cong X, Wei L
Experimental and therapeutic medicine 2017 Aug;14(2):1846-1852
Experimental and therapeutic medicine 2017 Aug;14(2):1846-1852
Antithymocyte Globulin at Clinically Relevant Concentrations Kills Leukemic Blasts.
Dabas R, Lee R, Servito MT, Dharmani-Khan P, Modi M, van Slyke T, Luider J, Durand C, Larratt L, Brandwein J, Morris D, Daly A, Khan FM, Storek J
Biology of blood and marrow transplantation : journal of the American Society for Blood and Marrow Transplantation 2016 May;22(5):815-24
Biology of blood and marrow transplantation : journal of the American Society for Blood and Marrow Transplantation 2016 May;22(5):815-24
Single-cell profiling of human megakaryocyte-erythroid progenitors identifies distinct megakaryocyte and erythroid differentiation pathways.
Psaila B, Barkas N, Iskander D, Roy A, Anderson S, Ashley N, Caputo VS, Lichtenberg J, Loaiza S, Bodine DM, Karadimitris A, Mead AJ, Roberts I
Genome biology 2016 May 3;17:83
Genome biology 2016 May 3;17:83
A novel autosomal recessive TERT T1129P mutation in a dyskeratosis congenita family leads to cellular senescence and loss of CD34+ hematopoietic stem cells not reversible by mTOR-inhibition.
Stockklausner C, Raffel S, Klermund J, Bandapalli OR, Beier F, Brümmendorf TH, Bürger F, Sauer SW, Hoffmann GF, Lorenz H, Tagliaferri L, Nowak D, Hofmann WK, Buergermeister R, Kerber C, Rausch T, Korbel JO, Luke B, Trumpp A, Kulozik AE
Aging 2015 Nov;7(11):911-27
Aging 2015 Nov;7(11):911-27
Generation of functional, antigen-specific CD8+ human T cells from cord blood stem cells using exogenous Notch and tetramer-TCR signaling.
Fernandez I, Ooi TP, Roy K
Stem cells (Dayton, Ohio) 2014 Jan;32(1):93-104
Stem cells (Dayton, Ohio) 2014 Jan;32(1):93-104
Inhibitory Receptor Expression Depends More Dominantly on Differentiation and Activation than "Exhaustion" of Human CD8 T Cells.
Legat A, Speiser DE, Pircher H, Zehn D, Fuertes Marraco SA
Frontiers in immunology 2013;4:455
Frontiers in immunology 2013;4:455
Development of Norwalk virus-specific monoclonal antibodies with therapeutic potential for the treatment of Norwalk virus gastroenteritis.
Chen Z, Sosnovtsev SV, Bok K, Parra GI, Makiya M, Agulto L, Green KY, Purcell RH
Journal of virology 2013 Sep;87(17):9547-57
Journal of virology 2013 Sep;87(17):9547-57
Hypermutation of the inactive X chromosome is a frequent event in cancer.
Jäger N, Schlesner M, Jones DT, Raffel S, Mallm JP, Junge KM, Weichenhan D, Bauer T, Ishaque N, Kool M, Northcott PA, Korshunov A, Drews RM, Koster J, Versteeg R, Richter J, Hummel M, Mack SC, Taylor MD, Witt H, Swartman B, Schulte-Bockholt D, Sultan M, Yaspo ML, Lehrach H, Hutter B, Brors B, Wolf S, Plass C, Siebert R, Trumpp A, Rippe K, Lehmann I, Lichter P, Pfister SM, Eils R
Cell 2013 Oct 24;155(3):567-81
Cell 2013 Oct 24;155(3):567-81
Targeting CDK1 promotes FLT3-activated acute myeloid leukemia differentiation through C/EBPα.
Radomska HS, Alberich-Jordà M, Will B, Gonzalez D, Delwel R, Tenen DG
The Journal of clinical investigation 2012 Aug;122(8):2955-66
The Journal of clinical investigation 2012 Aug;122(8):2955-66
Multiparametric flow cytometry for identification and fluorescence activated cell sorting of five distinct B-cell subpopulations in normal tonsil tissue.
Kjeldsen MK, Perez-Andres M, Schmitz A, Johansen P, Boegsted M, Nyegaard M, Gaihede M, Bukh A, Johnsen HE, Orfao A, Dybkaer K
American journal of clinical pathology 2011 Dec;136(6):960-9
American journal of clinical pathology 2011 Dec;136(6):960-9
CD38 binding to human myeloid cells is mediated by mouse and human CD31.
Horenstein AL, Stockinger H, Imhof BA, Malavasi F
The Biochemical journal 1998 Mar 15;330 ( Pt 3):1129-35
The Biochemical journal 1998 Mar 15;330 ( Pt 3):1129-35
No comments: Submit comment
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
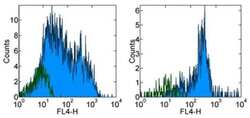
- Experimental details
- Staining of normal human peripheral blood cells with Mouse IgG1 K Isotype Control Alexa Fluor® 700 (Product # 56-4714-80) (open histogram) or Anti-Human CD38 Alexa Fluor® 700 (filled histogram). Cells in the lymphocyte (left) and monocyte (right) gates were used for analysis.
- Conjugate
- Near infrared dye
Supportive validation
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Near infrared dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Near infrared dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 4. IFNgamma is required for development of T-bet hi B DN cells and regulates ASC formation and recovery. ( a ) Ingenuity Pathway Analysis (IPA) to identify predicted upstream direct and indirect regulators of the HD B DN Be1 transcriptome. IPA performed using the 427 DEG (B DN Be1 over B DN Be2; FDR < 0.05) identified in the RNA-seq analysis described in Figure 3b . The predicted activation state (z-score of B DN Be1 over B DN Be2) of each regulator/signaling pathway is shown as bar color (orange, activated; blue, inhibited) with predicted upstream regulators sorted in order of significance (overlap P value). Regulators with an overlap P -value
- Conjugate
- Near infrared dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 5. Temporally distinct regulation of T-bet hi IRF4 int pre-ASC and ASC development by IFNgamma, R848 and IL-21. Cartoon ( a ) depicting stimulation of CTV-labeled HD B N cells for 3 days with anti-Ig, R848, IL-21 and IFNgamma (Step 1). Cells were washed and re-cultured for 3 days with R848, IFNgamma, and IL-21 (Step 2, +,+ condition) or individual stimuli were included in Step 1 only (+,- condition) or in Step 2 only (-,+ condition). Cells from day 6 cultures containing IFNgamma ( b-e ), R848 ( f-i ) or IL-21 ( j-m ) in Step 1, Step 2 or both steps were analyzed to determine ASC frequencies ( b, f, j ), ASC recovery ( c, g, k ), cell division ( d, h, l ) and total cell recovery ( e, i, m ). Summary of data ( n ) showing that ASC development and recovery from T-bet hi IRF4 int B DN pre-ASCs requires early IFNgamma, R848 and BCR ''priming'' signals and late R848 and IL-21 proliferation and differentiation signals. See Figure 5-figure supplement 1 for representative flow cytometry plots from each culture showing T-bet hi IRF4 int B DN cells on day 3, CD38 hi CD27 + ASCs on day 6 and CTV dilution on day 6. Data are representative of >=3 experiments. The percentage of cells in each division, the frequency of ASCs and cell recovery (total and ASCs) are shown as the mean +-SD of cultures containing purified B N cells from 3 independent healthy donors. All statistical analyses were performed using one-way ANOVA with Tukey''s multiple comparison test. P values *
- Conjugate
- Near infrared dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6-figure supplement 2. Flow cytometric characterization of B cells activated during the early priming phase in the presence or absence of IFNgamma and IL-2. Day 6 Be.0, Be.IL2, Be.IFNgamma and Be.gamma2 cells were generated as described in Figure 6e . Representative flow cytometry plots from day 6 Be.0, Be.IFNgamma, Be.IL2 and Be.gamma2 cultures showing CD38 hi CD27 + ASCs ( a ) and CTV dilution ( b ).
- Conjugate
- Near infrared dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Figure 6. IFNgamma cooperates with R848, IL-2 and IL-21 to promote development and recovery of ASCs. ( a-d ) IFNgamma synergizes with subthreshold amounts of TLR7/8 ligand to induce proliferation and differentiation of B N cells. CTV-labeled HD B N cells were activated for 3 days (Step 1) with anti-Ig, IL-2, and increasing concentrations of R848 (as indicated) in the presence or absence of IFNgamma (10 ng/ml). Cells were washed and re-cultured for 3 additional days (Step 2) with IL-21 and the same concentration of R848 that was used in Step 1. B cell division was measured on day 6 in cultures that were activated with IFNgamma (green circles) or without IFNgamma (orange circles) in the presence of no R848 (0 mug/ml, ( a ), high dose R848 (10 mug/ml, ( b ) or low dose R848 (0.1 mug/ml, ( c ). The frequency of CD38 hi CD27 + ASCs ( d ) on day 6 is shown. ( e-i ) IFNgamma cooperates with IL-2 to promote ASC development and recovery. Cartoon ( e ) depicting CTV-labeled HD B N cells activated for 3 days (Step 1) with anti-Ig and R848 alone (Be.0); with anti-Ig +R848+IFNgamma (Be.IFNgamma); with anti-Ig +R848+IL-2 (Be.IL2); or with anti-Ig +R848+IFNgamma+IL-2 (Be.gamma2). Cells were then washed and recultured for an additional 3 days (Step 2) with R848 and IL-21. The percentage of cells that have undergone cell division ( f ), the total cell recovery ( g ), the ASC frequencies ( h ) and total ASCs recovered ( i ) from each day 6 culture are shown. ( j-k ) Early IFNgamma signals regu
- Conjugate
- Near infrared dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig 4 p16 INK4a is a functional barrier to EBV driven proliferation of lymphoblastoid cells. (A) Blueprint of the primary transcript and the spliced mRNA with the three exons of CDKN2A on chromosome 9 encoding the p16 INK4a protein. The target site of the RNP complex within the 1st exon (exon1alpha) (chr9:21,974,678-21,974,827) is shown. (B) Study of the biological effect of the CDKN2A knockout in a time course experiment. WT and p16 KO cells were mixed such that the fraction of the latter was in the order of 10 to 20%, when the cells were infected with WT or DeltaEBNA3C EBV strains. The knockout status of the CDKN2A gene was studied by next generation sequencing to analyze the CD46 locus of the mixed cell populations over time. The fraction of cells with a disabled CDKN2A gene increased in cells infected with DeltaEBNA3C EBV exceeding 80% after eight weeks, whereas the knockout status of CDKN2A in the population of cells infected with WT EBV did not show a clear trend. Results from two biological replicates are shown, additional replicates can be found in S4A Fig . (C) Cell numbers of four different B cell populations were plotted as a function of days post nucleofection (x-axis) versus the format of the cell culture vessel (y-axis) starting with a single well in a 48-well cluster plate. 2x10 6 B cells with an intact CDKN2A locus (WT cells) or cells with an edited CDKN2A gene (p16 KO cells) were infected with wild-type (WT) EBV (left panel) or DeltaEBNA3C EBV (right panel).
- Conjugate
- Near infrared dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image
- Experimental details
- NULL
- Conjugate
- Near infrared dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 1 Marker verification and validation of independent markers in BALL cells by immunofluorescence and flow cytometry. The expression of CD20 ( a - c ) and CD38 ( d - f ), were positive on BALL cell membranes. CD90f ( g - i ) and CD49f ( j , k ) were not expressed in BALL cells. DAPI indicates the cell nucleus. This finding was confirmed via the conducted flow cytometry analysis. Scale bar = 100 mum
- Conjugate
- Near infrared dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- Fig. 3 CD20 and CD38 verification and validation of independent markers in SSCs by immunofluorescence and flow cytometry. CD20 ( a - c ) and CD38 ( d - f ) were not expressed in SSCs. DAPI indicates the cell nucleus. This finding was confirmed via the conducted flow cytometry analysis. Scale bar = 100 mum
- Conjugate
- Near infrared dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
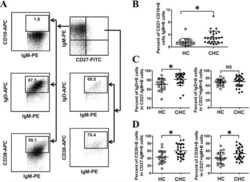
- Figure 7 Increased exhausted CD8 + T cells in convalescent patients. A. Expression of activation marker CD38 on CD8 + T cells (upper FACS panel). One exemplary dot plot is shown per study group. The bar diagram (lower panel) shows that CD38 expression was significantly higher on CD8 + T cells in convalescent COVID-19 + patients compared with HC. *P-value
- Conjugate
- Near infrared dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
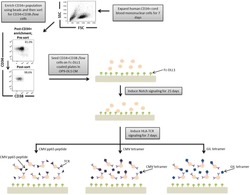
- A schematic overview of our experimental design to generate antigen-specific CD8+ T cells. CD34+ CB cells were expanded for 7 days and CD34+CD38-/low cells were isolated. After 7 days of CD34+ cell expansion, CD34+ cells were enriched using magnetic bead separation. The enriched cells were stained with anti-CD34 and anti-CD38 antibodies to isolate the CD34+CD38-/low HSC population. 81.0% of FSC versus SSC gated cells were CD34+CD38-/low. Gate was determined by the isotype staining control. Data are representative of at least six independent experiments. To induce Notch signaling and early T cell differentiation, CD34+CD38-/low cells were seeded onto DLL1-coated nontissue culture-treated plates. HLA-TCR signaling to direct antigen-specific differentiation was induced using CMVpp65 peptide or epitope-loaded HLA-A*0201 class I tetramers or GIL epitope-loaded HLA-A*0201 class I tetramers. Abbreviations: CM, conditioned medium; CMV, cytomegalovirus; FSC, Forward Scatter; GIL, Influenza M1 virus; HLA, Human leukocyte antigen; SSC, Side Scatter; TCR, T cell receptor.
- Conjugate
- Near infrared dye
- Submitted by
- Invitrogen Antibodies (provider)
- Main image

- Experimental details
- NULL
- Conjugate
- Near infrared dye
 Explore
Explore Validate
Validate Learn
Learn Flow cytometry
Flow cytometry